Vinegar in Ultrasonic Cleaners: The Complete Usage Guide
Browse Volume:32 Classify:Support
Ultrasonic cleaners have transformed how people tackle stubborn dirt and grime on everything from jewelry to mechanical parts. These machines create millions of microscopic bubbles that scrub surfaces with incredible precision. The question of what cleaning solution works best continues to spark debate among users. Vinegar, a household staple known for its cleaning properties, often enters this conversation.
Many ultrasonic cleaner owners wonder whether they can skip expensive commercial solutions in favor of affordable vinegar. The acidic nature of vinegar makes it effective for certain cleaning tasks, but compatibility concerns with both the machine and items being cleaned require careful consideration. Getting this decision right protects your equipment investment while achieving sparkling clean results.
The answer involves more nuance than a simple yes or no. Vinegar can work beautifully in ultrasonic cleaners for specific applications when used correctly. However, improper use risks damaging both your ultrasonic unit and the items you’re trying to clean. Understanding the chemistry, proper dilution, compatible materials, and best practices ensures successful outcomes.
Understanding Ultrasonic Cleaning Technology
Before diving into vinegar specifics, grasping how ultrasonic cleaners function helps explain why solution choice matters so much. These devices aren’t just fancy vibrating baths. They create a complex physical and chemical cleaning environment.
How Ultrasonic Cleaners Work
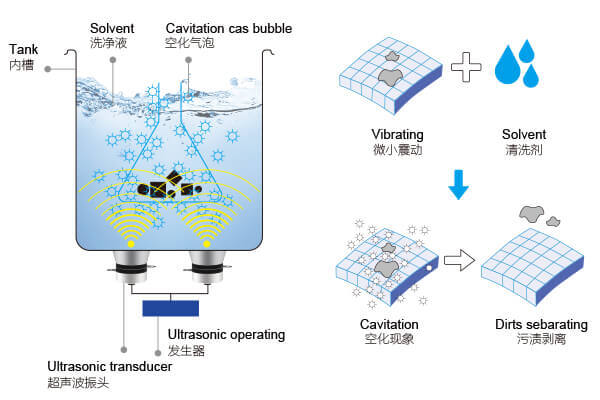
Ultrasonic cleaners generate high-frequency sound waves, typically ranging from 20 kHz to 200 kHz, which are beyond the range of human hearing. A transducer converts electrical energy into ultrasonic vibrations. These waves travel through the cleaning solution, creating alternating high-pressure and low-pressure cycles.
During low-pressure cycles, tiny vacuum bubbles form throughout the liquid. When pressure increases again, these bubbles violently collapse in a process called cavitation. Each bubble implosion creates a microscopic jet of liquid that impacts surfaces with surprising force. Millions of these bubble collapses per second provide intensive scrubbing action that reaches into crevices impossible to clean manually.
The Principle Behind Ultrasonic Cleaning
The cleaning solution itself carries away dislodged contaminants. The liquid medium also conducts ultrasonic waves throughout the tank. Without adequate liquid, the process simply doesn’t work. The chemical properties of that liquid significantly influence cleaning effectiveness.
The Role of Cleaning Solutions
Water alone provides the medium for cavitation but offers limited cleaning power. Adding solutions with specific chemical properties enhances the process. Surfactants reduce surface tension, allowing bubbles to form more readily. Solvents dissolve oils and greases. Acids neutralize alkaline residues and dissolve mineral deposits.
Different cleaning challenges require different chemical approaches. Removing oil from metal parts calls for degreasing agents. Eliminating mineral buildup benefits from acidic solutions. Delicate items need gentle, pH-neutral formulas. The solution you choose should match both the contaminant type and the material being cleaned.
Temperature also plays a crucial role. Most ultrasonic cleaners feature heating elements because warm solutions clean more effectively. Heat increases molecular activity, making chemical reactions proceed faster. The combination of heat, chemistry, and cavitation creates powerful cleaning synergy.
Can You Safely Use Vinegar in Ultrasonic Cleaners?
The compatibility question depends on multiple factors including your machine type, the items you’re cleaning, and how you prepare the vinegar solution. Blanket statements don’t serve anyone well. Let’s break down the specifics.
The Short Answer
Yes, you can use vinegar in most ultrasonic cleaners, but only when properly diluted and with compatible materials. Never use undiluted vinegar, as the strong acid can damage machine components and items being cleaned. White distilled vinegar diluted with water provides a safe, effective cleaning solution for many applications.
The key word is “diluted.” Straight vinegar contains about five percent acetic acid, which sounds mild but becomes aggressive in the intense environment of ultrasonic cavitation. The violent bubble collapses concentrate the acid’s effects, making dilution essential for safe use.
Most manufacturers don’t explicitly forbid vinegar use, but they recommend pH-neutral solutions to protect machine longevity. Using vinegar occasionally poses minimal risk. Regular use requires understanding your machine’s construction and taking protective measures.
Types of Vinegar and Their Properties
White distilled vinegar represents the best choice for ultrasonic cleaning. This clear, refined vinegar contains five to seven percent acetic acid with minimal other compounds. Its consistent composition makes results predictable. The absence of coloring agents prevents staining.
Apple cider vinegar, while popular for culinary and health uses, isn’t ideal for ultrasonic cleaners. The amber color can leave residues. Natural sugars and solids may create deposits in your machine. The slightly different acidity profile offers no cleaning advantages over white vinegar.
Cleaning vinegar, sold specifically for household cleaning tasks, contains up to twenty percent acetic acid. This stronger formulation works faster but requires even more dilution in ultrasonic applications. The increased acid concentration raises corrosion risks, making it suitable only for very short cleaning cycles on acid-resistant materials.
Balsamic, wine, and flavored vinegars have no place in ultrasonic cleaners. Their complex compositions, colors, and particulates cause more problems than they solve. Stick with plain white distilled vinegar for all ultrasonic cleaning applications.
Benefits of Using Vinegar as a Cleaning Solution
Understanding vinegar’s advantages helps explain its popularity among ultrasonic cleaner users. These benefits extend beyond simple cost savings, though that factor certainly matters.
Natural Acid Power
Vinegar’s acetic acid excels at dissolving mineral deposits, rust, and alkaline residues. Hard water stains vanish when exposed to acidic solutions. Tarnish on certain metals responds well to gentle acid treatment. Calcium buildup that resists other cleaners often surrenders to vinegar.
The acid neutralizes alkaline contaminants through straightforward chemistry. Many household and industrial soils contain alkaline compounds. An acidic cleaning solution balances the pH, breaking down these substances. The ultrasonic action distributes the acid throughout every surface contour, ensuring complete contact.
Vinegar also possesses mild antibacterial properties. While not a registered disinfectant, it reduces bacterial populations on cleaned items. This quality makes vinegar solutions particularly appealing for items like dental appliances or kitchen tools where sanitation matters.
Cost-Effectiveness
A gallon of white distilled vinegar costs a few dollars at most grocery stores. Commercial ultrasonic cleaning solutions often run ten to twenty times more expensive. When diluted properly, that gallon of vinegar produces many gallons of usable cleaning solution.
For home users running occasional cleaning cycles, cost differences multiply dramatically. Someone cleaning jewelry weekly might spend fifty dollars annually on commercial solutions. The same person using diluted vinegar spends perhaps five dollars for the entire year. Professional users with larger tanks and frequent cycles see even greater savings.
Budget considerations matter more in some situations than others. Hobbyists and home users appreciate the economy. Small businesses benefit from reduced operating costs. Industrial applications with specialized needs might justify premium solutions, but even they often use vinegar for certain tasks.
Environmental Advantages
Vinegar biodegrades completely and rapidly. Flushing diluted vinegar solutions down drains poses no environmental hazard. Municipal water treatment systems handle acetic acid easily. Compare this to commercial solutions containing synthetic surfactants, phosphates, or petroleum-derived solvents that stress wastewater systems.
Manufacturing vinegar produces minimal environmental impact compared to complex chemical solutions. The fermentation process that creates vinegar from ethanol occurs naturally. No exotic ingredients or energy-intensive synthesis steps enter the picture. Even the packaging tends toward simple recyclable containers.
For environmentally conscious users, vinegar represents the greenest ultrasonic cleaning option available. You maintain cleaning power while minimizing ecological footprint. This choice aligns with broader sustainability goals without sacrificing results.
Proper Vinegar Dilution Ratios
Getting dilution right makes the difference between safe, effective cleaning and potential damage. Too weak, and cleaning suffers. Too strong, and you risk corroding items or machine components. Follow these guidelines for optimal results.
Standard Dilution Guidelines
A general-purpose vinegar solution uses one part white distilled vinegar to three parts water. This creates roughly 1.25 to 1.75 percent acetic acid concentration, depending on your vinegar’s initial strength. This dilution safely cleans most compatible materials without excessive aggressiveness.
For light cleaning tasks like removing fingerprints from eyeglasses or freshening jewelry, dilute further to one part vinegar to five parts water. This gentler solution still provides adequate cleaning power with even greater safety margins. The ultrasonic action compensates for the reduced chemical strength.
Stubborn deposits might justify stronger solutions up to one part vinegar to one part water, but never exceed this fifty-fifty ratio. Use stronger concentrations only for very short cycles, typically under five minutes. Always test on inconspicuous areas first when using stronger solutions.
Measure accurately rather than estimating. Eyeballing ratios leads to inconsistent results and potential problems. Use measuring cups or marked containers to ensure proper proportions every time. Consistency matters in achieving predictable outcomes.
Adjusting Strength for Different Items
Delicate items like silver-plated jewelry or soft metals require weaker solutions around one part vinegar to six or eight parts water. The thin plating on silver-plated items can dissolve in stronger acids. Metals like aluminum show sensitivity to acids and benefit from gentler formulations.
Heavily soiled industrial parts might tolerate one-to-one vinegar-water mixtures for brief periods. Rust removal from steel tools, for example, works well with stronger solutions. Monitor closely and remove items as soon as cleaning completes to prevent over-exposure.
Items with mixed materials need solutions suitable for the most sensitive component. A watch with steel case and leather strap gets treated according to leather’s needs, which means no acidic solutions at all. Always consider the weakest link when determining appropriate solution strength.
Water Temperature Considerations
Warm solutions work more effectively than cold ones, but excessive heat causes problems. Most ultrasonic cleaners heat solutions to 130-150 degrees Fahrenheit. This temperature range optimizes cleaning without boiling or creating excessive evaporation.
Vinegar’s cleaning action accelerates at higher temperatures as molecular activity increases. The chemical reactions happen faster, meaning you can use slightly weaker solutions or shorter cycles when working with heated solutions. However, heat also increases corrosion potential on sensitive materials.
Never add vinegar to solutions above 160 degrees Fahrenheit. Acetic acid vapors become more concentrated at high temperatures, creating unpleasant fumes and potentially affecting your respiratory system. Let solutions cool slightly if your machine runs particularly hot.
Cold vinegar solutions work adequately for many tasks despite slower action. Room temperature cleaning just requires longer cycles to achieve the same results as heated solutions. This approach suits situations where you’re concerned about heat sensitivity of items being cleaned.
What Items Can Be Cleaned with Vinegar Solutions
Understanding which items safely tolerate vinegar solutions helps you make smart cleaning decisions. Material compatibility determines whether vinegar works or causes harm.
Metal Objects and Tools
Stainless steel tolerates diluted vinegar excellently. Kitchen utensils, tools, and hardware clean beautifully in vinegar solutions. The acid removes water spots, fingerprints, and light oxidation without damaging the corrosion-resistant surface. Stainless steel’s stability makes it ideal for vinegar cleaning.
Brass and copper respond well to vinegar when you want to remove tarnish. The acid dissolves the oxidation layer, restoring bright metal appearance. However, some brass items feature intentional patina that vinegar will strip away. Determine whether you want bright, shiny brass before proceeding.
Steel and iron parts clean effectively with vinegar, which removes rust and scale. Short cycles prevent excessive corrosion while eliminating surface contamination. Dry items immediately after cleaning and apply protective coatings to prevent new rust formation. Vinegar removes protective oils, leaving bare metal vulnerable.
Chrome-plated items handle diluted vinegar without issues. The hard chromium surface resists acid attack while benefiting from vinegar’s cleaning power. Bathroom fixtures, tools, and automotive trim pieces shine beautifully after vinegar ultrasonic treatment.
Eyeglasses and Optical Devices
Eyeglasses with glass or hard-coated plastic lenses clean perfectly in weak vinegar solutions. Use the gentlest dilution, one part vinegar to five or six parts water. The solution removes skin oils, dirt, and residues without harming lens coatings or frame materials.
Remove eyeglasses before the heated cycle completes if frames contain delicate components. Some nose pads or decorative elements might not tolerate extended heat exposure. The cleaning cycle itself requires only 10–30 seconds for eyeglasses.
Avoid vinegar solutions on anti-reflective coated lenses if the coating is old or damaged. Modern AR coatings tolerate mild acids, but vintage or degraded coatings might delaminate. When uncertain about coating quality, stick with plain water or commercial eyeglass solutions.
Binoculars, camera lenses, and other optical instruments generally should avoid ultrasonic cleaning entirely. The risk of damaging precision components outweighs cleaning benefits. If you must ultrasonically clean optical equipment, consult manufacturer guidelines and never use acidic solutions.
Jewelry Considerations
Gold jewelry tolerates diluted vinegar well. Pure gold doesn’t react with acetic acid. Gold alloys used in jewelry contain copper or silver that might tarnish slightly, but this reverses easily with gentle polishing. Remove jewelry as soon as cleaning completes to minimize exposure.
Sterling silver presents complications. Vinegar can clean silver but may also accelerate tarnishing if items remain in solution too long. Use very weak solutions and short cycles of three minutes or less. Rinse immediately and dry thoroughly to prevent tarnish formation.
Never use vinegar on pearls, opals, turquoise, or other porous gemstones. The acid penetrates these materials, causing permanent damage. Emeralds treated with oils, a common practice, also suffer from vinegar exposure which dissolves the protective oils.
Diamonds, rubies, and sapphires handle vinegar without concern. These hard, non-porous stones resist acid attack completely. However, verify that settings tolerate vinegar before cleaning valuable pieces. Glued components or soft metal settings might not survive the treatment.
Kitchen and Household Items
Glassware sparkles after ultrasonic cleaning with vinegar solutions. The acid dissolves hard water deposits and soap scum that dull glass surfaces. Wine glasses, vases, and glass food containers emerge crystal clear. Use standard dilution ratios and normal cycle times.
Ceramic and porcelain items clean beautifully with vinegar. The glazed surfaces resist acid while benefiting from its cleaning power. Coffee cups with stubborn stains, decorative ceramics with intricate details, and porcelain figurines all respond well to vinegar ultrasonic treatment.
Stainless steel flatware, serving pieces, and cookware accessories clean thoroughly in vinegar solutions. The combination of acid and cavitation removes baked-on residues and discoloration. Never ultrasonically clean items with wooden handles or heat-sensitive decorative elements.
Dental appliances like retainers or night guards benefit from gentle vinegar cleaning. Use very weak solutions, about one part vinegar to eight parts water. The mild antibacterial properties of vinegar complement the mechanical cleaning action. Always rinse thoroughly before wearing.
Items That Should Never Touch Vinegar
Knowing what to avoid prevents costly mistakes and destroyed valuables. Some materials simply don’t tolerate acidic solutions regardless of dilution or exposure time.
Delicate Gemstones
Pearls top the list of stones damaged by vinegar. The nacre coating that gives pearls their luster consists of calcium carbonate. Acids dissolve calcium carbonate, permanently ruining pearls. Even brief exposure causes irreversible pitting and dulling.
Opals contain significant water content within their structure. Acid exposure disrupts this internal moisture balance, causing crazing or cracking. The rainbow fire that makes opals beautiful disappears when the stone’s structure fails. Never risk opals in any acidic solution.
Turquoise, coral, and amber all suffer from acid exposure. These porous or organic materials absorb liquids readily. Vinegar penetrates deeply, altering color and structural integrity. The damage continues even after removing items from solution as absorbed acid keeps working.
Lapis lazuli contains calcite that reacts with acids, causing dulling and pitting. Malachite similarly features copper carbonate that vinegar attacks. When cleaning jewelry with mixed stones, identify every stone type before selecting a cleaning method.
Porous Materials
Natural stones like marble, limestone, and travertine contain calcium carbonate that vinegar dissolves. Countertop samples, decorative objects, or stone-inlaid jewelry suffer etching and dulling from acid contact. The damage appears as dull spots or rough patches where smooth polish existed.
Concrete and grout react similarly to natural stone. The cement binding these materials together contains alkaline compounds that acid neutralizes, weakening the structure. Never clean concrete items or grouted objects in vinegar solutions.
Unsealed wood absorbs vinegar, causing swelling, warping, and discoloration. Musical instruments, wooden jewelry boxes, or items with wooden components must avoid ultrasonic cleaning with any liquid. The wood grain raises and splits when repeatedly wetted and dried.
Leather and fabric cannot withstand ultrasonic cleaning in general, regardless of solution type. The mechanical action damages fibers while liquids cause staining and deterioration. Remove leather watchbands or fabric elements before cleaning other components of an item.
Certain Metal Types
Aluminum reacts with acids, developing dull gray spots or white powdery deposits. While aluminum resists many chemicals, acetic acid attacks the protective oxide layer. The ultrasonic action accelerates this reaction, causing rapid visible damage.
Cast iron requires special care. The seasoning layer that prevents rust and provides non-stick properties strips away in acidic solutions. Raw cast iron then rapidly develops rust in the presence of water and acid. Only clean cast iron with neutral solutions and immediately re-season.
Zinc and zinc-plated items suffer in vinegar. The acid dissolves zinc relatively quickly, leaving a rough, corroded surface. Galvanized steel, which features zinc coating, similarly deteriorates in acidic ultrasonic baths. Check plating types before cleaning hardware or fasteners.
Lead, pewter, and other soft metals can pit or corrode in vinegar. While less common in modern items, antique pieces might contain these materials. When uncertain about metal composition, avoid acidic solutions or test inconspicuous areas first.
Potential Risks and Machine Compatibility
Using vinegar affects your ultrasonic cleaner itself, not just the items being cleaned. Understanding these impacts helps you make informed decisions about solution selection and machine maintenance.
Tank Material Concerns
Most ultrasonic cleaners feature stainless steel tanks that tolerate diluted vinegar without issues. Stainless steel’s corrosion resistance protects against weak acids during normal use. However, prolonged contact with undiluted or highly concentrated vinegar can eventually pit even stainless steel.
Some budget ultrasonic cleaners use plastic tanks. Quality plastics resist acetic acid well, but cheaper materials might degrade over time. Look for tanks made from polypropylene or similar acid-resistant plastics. Avoid cleaners with unknown plastic compositions when planning regular vinegar use.
Aluminum tank coatings exist on some units, particularly older or very inexpensive models. These tanks absolutely should not contact vinegar solutions. The acid rapidly corrodes aluminum, ruining the tank and potentially contaminating items being cleaned. Check your manual for tank material specifications.
Always empty vinegar solutions immediately after use. Don’t let acidic solutions sit in your ultrasonic cleaner for hours or days. The prolonged exposure increases corrosion risk even on resistant materials. Rinse the tank with plain water after vinegar use to remove residual acid.
Seal and Gasket Effects
Rubber seals and gaskets in ultrasonic cleaners can deteriorate from repeated acid exposure. The acetic acid gradually breaks down rubber compounds, causing swelling, cracking, or loss of elasticity. Failed seals lead to leaks and potential electrical hazards.
Silicone seals resist acids better than natural rubber, but still suffer from extended exposure. Minimize contact by thoroughly rinsing the tank after vinegar use. Wipe visible seals with a damp cloth to remove any solution residue.
Some ultrasonic cleaners position heating elements or transducers with sealed connections that mustn’t fail. Acid infiltration into these areas can cause corrosion of electrical components. This creates safety hazards and expensive repairs or replacement needs.
Inspect seals periodically if you regularly use vinegar solutions. Look for swelling, cracking, or hardening that indicates deterioration. Replace compromised seals promptly to maintain machine integrity and safe operation. Preventive maintenance costs far less than emergency repairs.
Long-Term Machine Health
Manufacturers design ultrasonic cleaners expecting neutral or mildly alkaline commercial solutions. Using acids like vinegar falls outside typical design parameters. While occasional use causes minimal problems, making vinegar your primary solution accelerates wear.
The transducer, which converts electrical energy to ultrasonic vibrations, can suffer from acid exposure over years. Corrosion of the transducer face reduces cleaning efficiency and eventually causes failure. These components cost significant money to replace.
Heating elements face similar risks. Exposed metal surfaces gradually corrode in acidic environments. Corrosion creates rough spots that accumulate mineral deposits, reducing heating efficiency. Eventually the element fails completely, requiring professional repair.
Balance vinegar’s benefits against these long-term considerations. For users who clean occasionally, the minimal wear proves acceptable. Professional users running daily cycles might prefer commercial solutions formulated for ultrasonic equipment despite higher costs.

Granbo ultrasonic cleaners
Comparing Vinegar to Commercial Solutions
Understanding how vinegar stacks up against purpose-made ultrasonic solutions helps you decide when each option makes sense. Neither represents the perfect choice for every situation.
Performance Differences
Commercial ultrasonic solutions contain surfactants that reduce surface tension more effectively than vinegar alone. Lower surface tension allows cavitation bubbles to form more readily and collapse more violently. This translates to slightly faster, more thorough cleaning in many applications.
Specialty solutions target specific contaminants. Degreasing formulas contain solvents that dissolve oils better than vinegar. Jewelry cleaners include anti-tarnish compounds and brighteners. Metal cleaners feature corrosion inhibitors alongside cleaning agents. These targeted formulas outperform generic vinegar solutions for specialized tasks.
However, vinegar’s acid power excels at mineral deposit removal. Commercial solutions often handle oils and general grime better, but struggle with hard water stains or rust that vinegar tackles easily. For these specific challenges, vinegar actually outperforms many commercial products.
pH-neutral commercial solutions clean gently without any corrosion concerns. This safety margin matters for valuable or irreplaceable items. The extra cost buys peace of mind that vinegar cannot provide for delicate pieces.
Cost Analysis
A typical bottle of commercial ultrasonic solution costs fifteen to thirty dollars and makes several gallons of diluted cleaning mixture. A gallon of white vinegar costs three to five dollars and makes comparable quantities at proper dilutions. The math clearly favors vinegar for budget-conscious users.
Professional users face different economics. High-volume operations buying commercial solutions in bulk pay dramatically less per gallon. Specialized formulas that increase efficiency and reduce cycle times save labor costs. These factors can justify premium solution costs in commercial settings.
Consider the value of items being cleaned. Using five-dollar vinegar on a hundred-dollar watch makes economic sense. Using it on a five-thousand-dollar diamond ring seems penny-wise and pound-foolish. Match solution costs to item values and cleaning requirements.
When Commercial Solutions Win
Extremely delicate items justify commercial solutions regardless of cost. Antique jewelry, precision instruments, or valuable collectibles deserve the gentlest cleaning chemistry available. The safety margin of pH-neutral commercial formulas provides insurance against accidents.
Mixed-material items clean better in specialized solutions formulated to handle diverse components safely. A necklace combining pearls, gold, and crystal benefits from jewelry-specific solutions that safely clean all materials simultaneously. Vinegar would destroy the pearls while cleaning the metal.
Time-sensitive situations favor fast-acting commercial solutions. Professional jewelry stores cleaning customer pieces need quick turnaround. The marginal speed advantage of surfactant-enhanced commercial solutions matters in business contexts even if vinegar would eventually achieve similar results.
Warranty considerations influence solution choice. Some ultrasonic cleaner manufacturers specify approved solutions in warranty terms. Using vinegar might void coverage on expensive machines. Check warranty documents before deviating from manufacturer recommendations.

Ultrasonic Cleaning Agents
Alternative Natural Cleaning Solutions
Vinegar isn’t the only household option for ultrasonic cleaning. Several other common substances provide effective, economical cleaning power.
Baking soda dissolved in water creates a mildly alkaline solution excellent for greasy or oily contaminants. Use one tablespoon per quart of water. This solution works particularly well on kitchen items or tools covered in oil-based grime. Baking soda won’t harm most materials, though it may dull some metal finishes.
Lemon juice functions similarly to vinegar with comparable acidity. The citric acid provides equivalent cleaning power against mineral deposits. However, lemon juice costs more than vinegar and may leave sticky residues if not thoroughly rinsed. Most users find no compelling reason to choose lemon juice over vinegar.
Dish soap mixed with water creates a gentle cleaning solution suitable for delicate items. Use just a few drops per quart to avoid excessive foaming. Soap solutions clean effectively without the corrosion concerns of acidic options. This represents the safest choice for valuable or uncertain items.
Ammonia diluted in water tackles stubborn grime and provides degreasing action. However, ammonia fumes irritate respiratory systems and mixing with other cleaners creates dangerous chemical reactions. Unless you have specific needs that ammonia addresses, simpler alternatives work better for most users.
Tips for Maximum Effectiveness
Learning from experienced ultrasonic cleaner users helps avoid common pitfalls and achieve better results. These insights come from both professional applications and serious hobbyist experience.
Test solutions on inconspicuous areas first when cleaning valuable or uncertain items. This simple precaution catches compatibility problems before they ruin entire pieces. Choose hidden spots like the inside of ring bands or the back of pendants for testing. Wait several hours after testing to ensure delayed reactions don’t occur.
Maintain solution temperature consistently throughout cleaning cycles. Temperature fluctuations reduce cleaning efficiency and can cause thermal stress in some materials. Pre-heat solutions before adding items when possible. Monitor temperature displays and adjust settings as needed.
Degass new solutions before use by running a cycle without items present. Fresh solutions, especially those made with cold tap water, contain dissolved gases that interfere with cavitation. Running a short empty cycle releases these gases, optimizing bubble formation for subsequent cleaning cycles.
Position items carefully in the basket to maximize exposure to cavitation. Place objects vertically when possible so surfaces face perpendicular to the tank bottom. Avoid nesting pieces inside each other, which shields interior surfaces from cleaning action. Give each item adequate space for solution circulation.
Change solutions when they become visibly dirty. Contaminated solutions redeposit grime onto supposedly clean items. The cleaning power also diminishes as active ingredients become spent. Fresh solutions clean faster and better than reused ones regardless of chemical type.
Common Mistakes to Avoid
Understanding frequent errors helps you sidestep problems that plague less experienced users. These mistakes range from minor inconveniences to serious damage.
Using undiluted vinegar represents perhaps the most common error. Straight vinegar seems like it should clean better, but the excessive acidity causes rapid corrosion and damages delicate items. Always dilute according to guidelines. Stronger rarely equals better with ultrasonic cleaning.
Leaving items in solution after cycles complete allows unnecessary acid exposure. The damage accumulates gradually rather than occurring dramatically. Form the habit of promptly removing items as soon as the timer signals completion. This simple practice prevents many problems.
Mixing different cleaning solutions creates unpredictable chemical reactions. Combining vinegar with ammonia, for example, produces hazardous compounds. Use one solution type per session. Empty and rinse thoroughly before switching to different chemicals.
Overloading the ultrasonic cleaner reduces effectiveness dramatically. Crowded baskets prevent proper cavitation around items. Pieces touching each other shield surfaces from cleaning action. Clean smaller batches more thoroughly rather than large batches poorly.
Neglecting post-cleaning rinses leaves chemical residues that continue reacting with materials. This oversight causes slow-developing problems like tarnish or corrosion that appear days later. Always rinse thoroughly regardless of cleaning solution used.
 Granbo Sonic
Granbo Sonic















